The Porphyrin Thread: how a single ancient molecule connects the green leaf to the red blood
Article 1 of a series: Dialogues with Claude.ai
A note on deep-time science:
The events described in this article reach back billions of years, well beyond any direct fossil record. Deep-time science operates by inference, laboratory simulation, isotopic analysis, and analogy — not by observation. Where I write that something ‘happened,’ I mean that the best current evidence suggests it happened. The honest word is always ‘inferred.’ I ask the reader to hold that qualifier in mind throughout.
A conversation I was having with Claude.ai about the similarities in chlorophyll and hemoglobin took an unexpected turn. What began as a straightforward question about two molecules ended somewhere I had not anticipated: at the edge of what we mean by mind and by self. The path from that question leads to another, single, molecule — one of the most ancient still in active use on Earth — and following it enhanced my fellow-feeling with the avocado plant sitting for years beside my front room window.
This is the first in a series of articles arising from this conversation. The science leads; the philosophy follows. I have intended to be accurate about what is established, what is inferred, and what remains open.
One Ring, Two Purposes
Chlorophyll and hemoglobin look, at first glance, like unrelated molecules doing unrelated jobs in unrelated kingdoms of life. One makes leaves green and captures sunlight in plants. The other makes blood red and carries oxygen in animals. Yet at their core they share an identical structural foundation: the porphyrin ring — a large, flat, cyclic molecule built from four linked pyrrole units with a metal atom at its center.
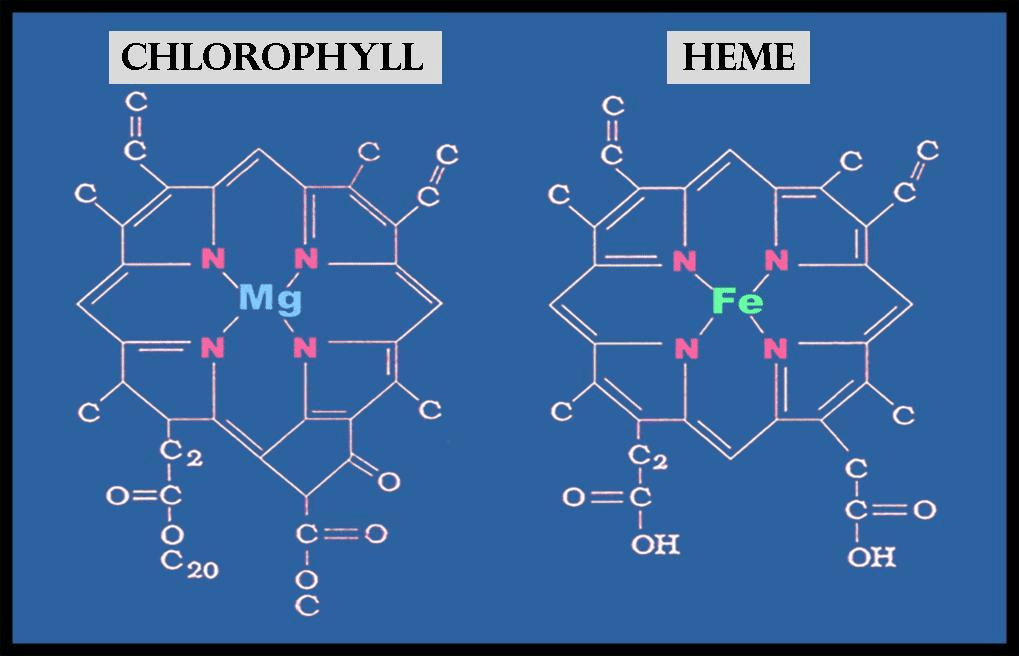
The difference that matters most is which metal sits at that center. Chlorophyll holds magnesium. Hemoglobin holds iron. Swap the metal, change the surrounding protein, and you move from capturing the sun’s energy to carrying it through an animal’s body. The same molecular scaffold underlies two of life’s most fundamental processes — a convergence whose deeper meaning we will return to.
Both molecules are also, at bottom, about electron behavior. Chlorophyll uses light to move electrons, generating chemical energy. Hemoglobin uses iron’s electron configuration to hold and release a molecule — oxygen — that will eventually accept electrons in cellular respiration. They are, in a sense, two ends of the same electron economy.
The question of why the same scaffold appears in such different contexts leads directly into the deep history of life itself.
A Common Ancestry — With a Caveat
The shared porphyrin scaffold is almost certainly not coincidence. Both chlorophyll and hemoglobin are synthesized by living cells along the same biochemical pathway, diverging only at a late branch point — where iron is inserted for heme, magnesium for chlorophyll. This shared biosynthetic route is strong evidence that a single ancestral organism possessed porphyrin chemistry long before the divergence of plants and animals.
Heme-containing proteins — the cytochromes — are found in all three domains of life: Bacteria, Archaea, and Eukaryota. This universality points toward an origin in LUCA, the Last Universal Common Ancestor of all life, inferred to have existed perhaps 3.8 billion years ago.
And porphyrins themselves have been produced in laboratory simulations of prebiotic conditions, and found in carbonaceous meteorites — including the Murchison meteorite that fell in Australia in 1969. This suggests that porphyrin chemistry is not a biological invention but a feature of organic chemistry generally, arising wherever the right conditions exist.
Here the caveat is necessary: finding that porphyrins can form in a laboratory, or did form in a meteorite, is not the same as knowing they formed on early Earth in the way that led to life. The inference is reasonable — chemists find it more surprising that porphyrins would not form under prebiotic conditions than that they would — but it remains inference, not observation. The rock record of the first billion years of Earth’s history has been almost entirely destroyed by geological processes. What we know of that period we know obliquely.
With that caveat held in mind: the porphyrin ring may be older than life itself. Which raises the unsettling possibility — one we will pursue in a later article — that asking when life began is harder than it sounds, and that the boundary between living and non-living chemistry is not where we habitually draw it.
The Oxygen Catastrophe
For the first billion or more years of life, there was no free oxygen in the atmosphere. The early Earth was a reducing world — nitrogen, carbon dioxide, methane, ammonia — and all life was anaerobic. The living world at this stage consisted entirely of prokaryotes — single-celled organisms without a nucleus, the ancestors of today’s bacteria and archaea. To them, oxygen was not a resource but a poison.
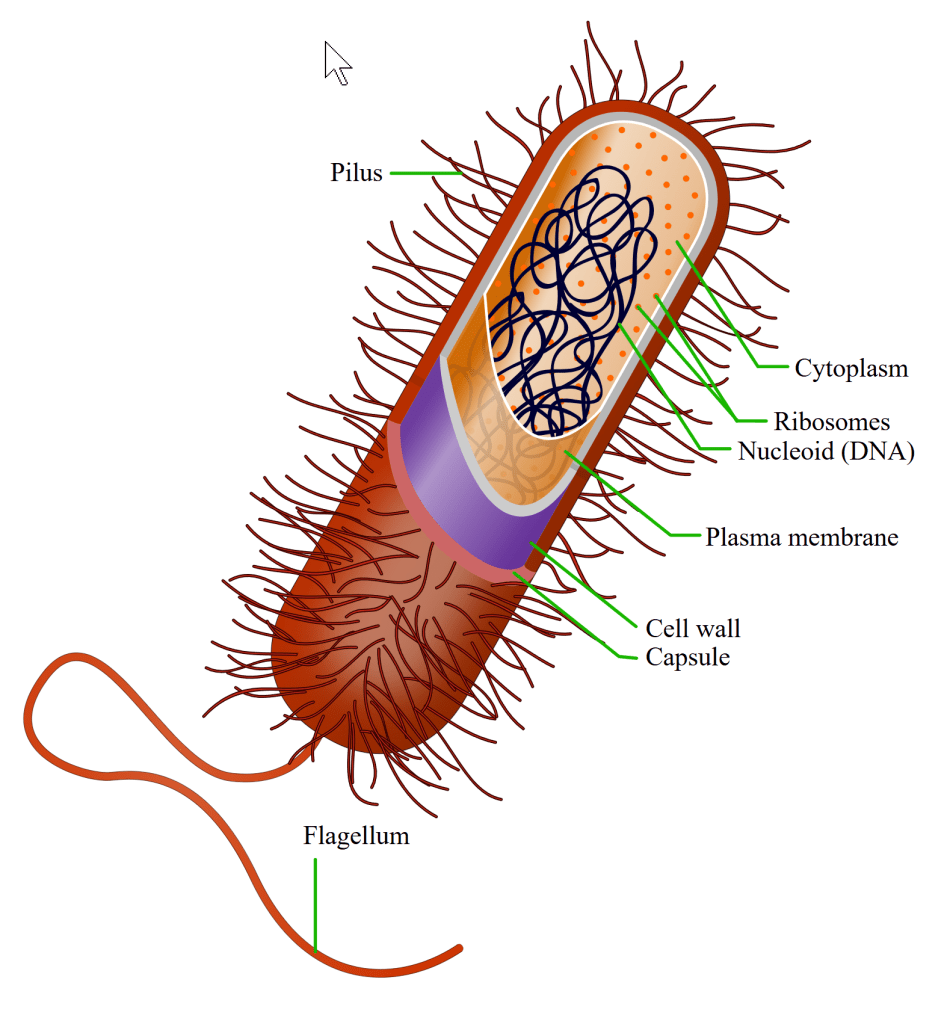
Diagram of a prokaryotic cell, a bacterium with a flagellum. (This image is made by Ali Zifan )
Around 2.7 billion years ago, by current inference, one lineage of prokaryotes — the cyanobacteria, themselves descended from earlier photosynthetic ancestors — developed chlorophyll-based photosynthesis through the slow accumulation of mutations in their porphyrin biosynthesis machinery. The end result was the ability to split water molecules using sunlight, releasing oxygen as a byproduct. Their chlorophyll-based machinery was the engine of what proved to be the most consequential transformation in the history of life. For hundreds of millions of years, the oxygen they produced was absorbed by dissolved iron in the oceans and by volcanic gases. Then, around 2.4 billion years ago, those chemical buffers saturated, and oxygen began accumulating in the atmosphere.
(The question of when and how the first photosynthetic organisms appeared reaches back into territory where the rock record is largely destroyed and certainty gives way to inference. It connects to the larger question of where life itself began — on Earth, in space, or at some boundary between chemistry and biology that may not be a boundary at all. Article 3 pursues this.)
The result was catastrophic for the anaerobic world — the largest extinction event in Earth’s history, invisible to us because it left no macroscopic fossils. Among the prokaryotes, variation in biochemical defenses meant that some were better able to tolerate the rising oxygen than others — not by design, but by the accident of existing chemistry. But for the survivors, it opened an extraordinary opportunity. Aerobic respiration — using oxygen as the terminal electron acceptor in metabolism — yields roughly eighteen times more energy per glucose molecule than anaerobic fermentation. Organisms able to exploit oxygen gained an enormous metabolic advantage.
At the center of this new aerobic metabolism were the cytochromes — heme proteins, porphyrin-based, already present before the catastrophe. The same heme scaffold that had managed electrons in the anaerobic world turned out to be well suited to managing oxygen. No one repurposed anything; the environment changed, and the molecules that happened to work in the new conditions were the ones that persisted and were refined. It is worth holding this in mind throughout: when we say a molecule was ‘repurposed’ or a bacterium ‘developed’ something, we are using convenient shorthand for a process in which science detects no conscious agent and requires none to explain what is observed. Chlorophyll produced the poison that became the resource; heme managed the resource that the poison had made available. One porphyrin creates the condition; another was already suited to exploit it.
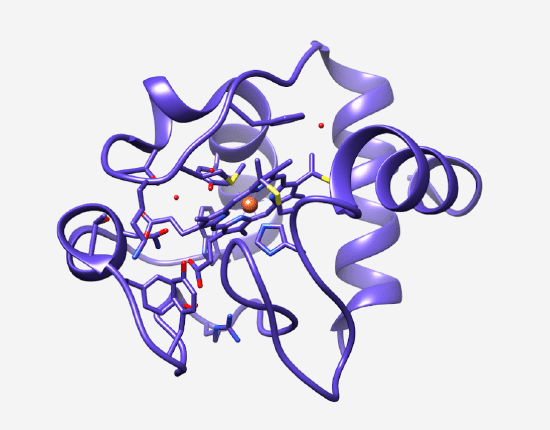
Cytochrome c: the heme group sits within a folded protein chain. This ancient molecule, present in virtually all living cells, carries the porphyrin ring that predates the divergence of all life on Earth.
When Life Swallowed Life
This chapter is the one I find most vertiginous, because it dissolves the boundary not just between plant and animal but between individual and community.
Around 2 billion years ago, a eukaryotic cell — a cell with a nucleus — engulfed an aerobic bacterium, an alpha-proteobacterium expert at oxygen metabolism. (How the nucleus itself arose is a separate and still-debated question, which we set aside here.) Instead of digesting it, the cell kept it alive. Over millions of generations, the bacterium lost its independence while the host gained vastly superior energy production. The bacterium became the mitochondrion — the energy-generating organelle present in virtually every complex cell alive today.
Every cell in your body contains what are, in a precise biological sense, domesticated bacteria. They are still bounded by a double membrane — the inner membrane is the original bacterial membrane, the outer is the remnant of the engulfing event. They still carry their own small circular genome, still divide by binary fission as bacteria do. Your mitochondria are not like bacteria. They are bacteria — or rather, what bacteria become after two billion years of enforced internal residence.
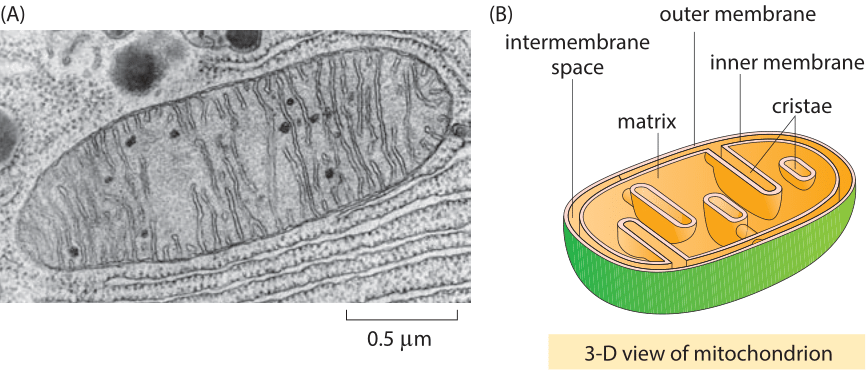
Image: https://book.bionumbers.org/
Mitochondria (the oval structure) photographed inside a cell by electron microscopy. Each is bounded by a double membrane — the outer a remnant of the ancient engulfment event, the inner the original bacterial membrane. These are not components that the cell invented; they are, in the precise biological sense, domesticated bacteria that have lived inside our cells for approximately two billion years. Every one of your cells contains hundreds of them.
A second endosymbiotic event followed, perhaps five hundred million years later. A eukaryotic cell — already containing mitochondria — engulfed a cyanobacterium. This too survived and became the chloroplast. The result was the first plant cell: a eukaryote running both porphyrin systems simultaneously — the ancient heme in its mitochondrial cytochromes and the magnesium porphyrin in its chloroplasts, cooperating in the same cytoplasm, descendants of the same prebiotic chemistry, now serving complementary functions in a single organism.
The biologist Lynn Margulis championed this theory of endosymbiosis in 1967, after it was rejected by some fifteen journals. It is now one of the most thoroughly confirmed theories in biology. The evidence is multiple and mutually reinforcing: the double membranes, the bacterial-sized genomes, the bacterial-type ribosomes, the fact that antibiotics targeting bacterial ribosomes also disrupt mitochondrial function.
Its implications go beyond cell biology. You are not, in any simple sense, an individual organism. You are a community — a eukaryotic cell lineage carrying captive bacteria in every cell, together with approximately 38 trillion bacterial cells in your gut and on your surfaces. The boundary of self is not where it appears to be.
What the Thread Reveals
Stand back and consider what this story has shown. A porphyrin ring, inferred to have arrived before any cell existed, was incorporated into the first life and carried forward through every subsequent lineage. It drove the electron transfer of the anaerobic world. When chlorophyll-based photosynthesis emerged in cyanobacteria and transformed the atmosphere, the same heme scaffold — already present — proved suited to managing the new oxygen economy. When endosymbiosis produced the complex cell, both porphyrin systems were brought inside — cooperating ever since in every plant cell on Earth.
The division between plant and animal, at the molecular level, is a late and shallow variation on a theme established billions of years before either kingdom existed. The green leaf and the red blood are not opposites. They are inflections of the same ancient chemistry.
And the story consistently dissolves the boundaries we are inclined to draw: between producer and consumer, between self and other, between individual and community. What appears to be a unit is always, on closer inspection, a community with a history. What appears to be a boundary is always a membrane with traffic flowing both ways.
—

The next article in this series asks where this picture leads philosophically. If the boundary between individual and community is permeable at the cellular level — if you are, literally, a community of former strangers now operating as one — what does this suggest about the nature of experience itself? Does the cooperative, integrated character of the living cell tell us something about mind?
Does mind go all the way down?
Ron Pavellas
Stockholm, April 2026
Here are the Articles in this Series:
Dialogue 1. Why Aren’t You a Plant?
Dialogue 2. Does Mind Go All the Way Down?
Dialogue 3. Did Life Begin Here at All?
Dialogue 4a. Did I Choose Any of This?
Dialogue 4b. Who Am I to Propound Upon Free Will?
Final Dialogue: 5. The Flowering Universe
Leave a comment